The hydrophobic lipid bilayer forms a barrier between the interior and the exterior of the cell. Thus, the transport of polar molecules needs to be modulated. Polar molecules, such as water and certain proteins, and ions need a transporter in the plasma membrane to cross. This is the function of membrane proteins.
Why is it important that membrane lipids are amphipathic?
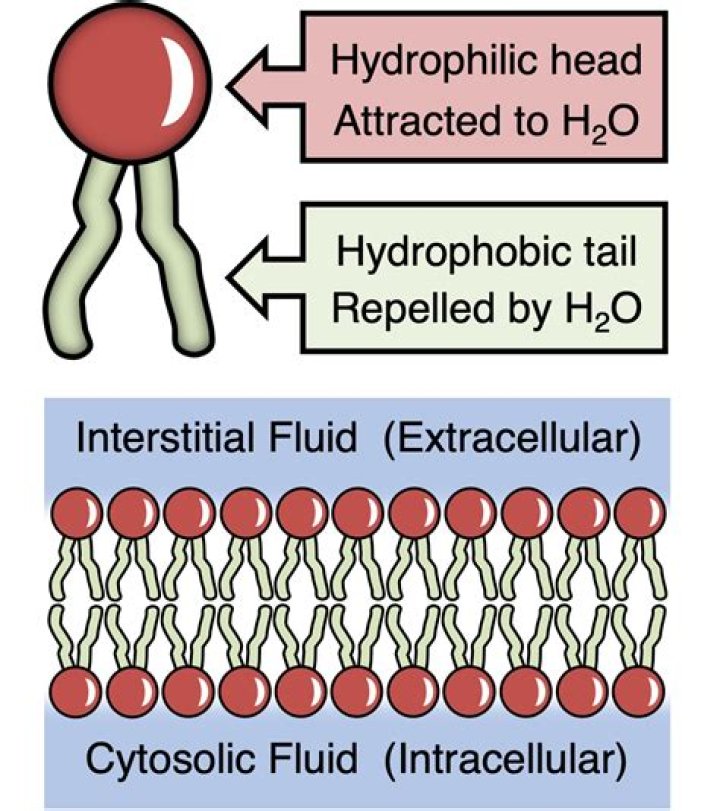
Membrane lipids are amphipathic, which means that they have a polar or hydrophilic end and a non-polar or hydrophobic end. … Furthermore, the hydrophobic interior of membranes serves as a barrier to water soluble molecules but allows certain lipid soluble molecules to passively diffuse through.
What do amphipathic lipids do?
Amphipathic molecules serve several important biological roles. They are the primary component of the lipid bilayers that form membranes. … The body uses amphipathic molecules for digestion. Amphipaths are also important in the immune response.
How do lipids affect the cell membrane?
In addition to the barrier function, lipids provide membranes with the potential for budding, tubulation, fission and fusion, characteristics that are essential for cell division, biological reproduction and intracellular membrane trafficking.Why does the phospholipid of the cell membrane is considered as amphipathic?
Phospholipids are amphipathic because they have hydrophobic tails and hydrophilic heads. … This part of the molecule is hydrophobic and faces inward in the cell membrane to avoid contact with water. The heads however, are made of a glycerol molecule bound to a phosphate group.
What does it mean for a molecule to be amphipathic?
adjective. Chemistry (of a molecule) having two different affinities, as a polar end that is attracted to water and a nonpolar end that is repelled by it.
Why are amphipathic molecules important constituents of membranes?
In biology, amphipathic molecules are important in the formation of biological membranes and micelles. Through them, the plasma membrane, in particular, is able to create an effective selective barrier so that not all substances can enter or exit the cell. Instead, some of them need transport mechanisms.
Are lipids needed to form cell membranes?
The three main types of lipids are triacylglycerols (also called triglycerides), phospholipids, and sterols. … Phospholipids are crucial for building the protective barrier, or membrane, around your body’s cells. In fact, phospholipids are synthesized in the body to form cell and organelle membranes.How does lipids affect membrane fluidity?
One way to remember how different lipids affect membrane fluidity or rigidity is that lipids that can pack more tightly (like saturated fatty acids and sterols) make membranes more rigid and stronger, but less fluid.
What is the difference between amphoteric and amphipathic?is that amphoteric is (chemistry) having the characteristics of both an acid and a base, and capable of reacting as either; amphiprotic while amphipathic is (chemistry) describing a molecule, such as a detergent, which has both hydrophobic and hydrophilic groups.
Article first time published onWhich membrane protein is amphipathic?
Membrane proteins (MPs) are amphipathic biomacromolecules with a central hydrophobic segment and two flanking hydrophilic moieties. Because of the presence of large hydrophobic surfaces, these membrane macromolecules are normally located in membrane bilayers comprised of amphipathic lipids.
What is an amphipathic lipid cite an example?
Cholesterol, detergents, and phospholipids are an example of amphipathic molecules. Complete Answer: … – Most membrane lipids are amphipathic. The typical example is phospholipids found in the cell membrane. The hydrophilic region is also called the water-loving head and face outside.
Which one of the following is an amphipathic lipid?
The amphipathic molecule has several hydrophilic and hydrophobic parts. Some of the examples of amphipathic molecules include bile salts, surfactants, and phospholipids. So, the correct answer is, ‘(c) Phospholipids’.
Why are lipids hydrophobic?
Lipids are mainly composed of carbon and hydrogen atoms, and this hydrophobic (“water fearing”) nature of lipids is driven by the bonds between these many carbons and hydrogens. … Thus, long chains of carbon-hydrogens bonds form a nonpolar molecule.
Are amphipathic and amphiphilic the same thing?
is that amphipathic is (chemistry) describing a molecule, such as a detergent, which has both hydrophobic and hydrophilic groups while amphiphilic is (chemistry|of a molecule) being a detergent: having both hydrophilic and hydrophobic (or lipophilic) groups.
What factors affect the fluidity of the cell membrane?
- Factor #1: The length of the fatty acid tail. The length of the fatty acid tail impacts the fluidity of the membrane. …
- Factor #2: Temperature. …
- Factor #3: Cholesterol content of the bilayer. …
- Factor #4: The degree of saturation of fatty acids tails.
What lipid makes the membrane more fluid?
Another type of lipid in the cell membrane is cholesterol that makes the membrane more fluid and adds to its flexibility.
Why are lipids and proteins free to move laterally in membranes?
Why are lipids and proteins free to move laterally in membranes? There are only weak hydrophobic interactions in the interior of the membrane.
Does lipids exhibit increased catalytic activity?
Explanation: Fatty acids are long-chain hydrocarbons and are the simplest form of lipids. … Explanation: Exhibit increased catalytic activity is incorrect as all other options are correct. Lipids take part in a structural component of the membrane of animals, plants, or bacteria where it has protective functions.
What is the manufacture of lipids required for making cell membrane?
The smooth endoplasmic reticulum functions in many metabolic processes. It synthesizes lipids, phospholipids as in plasma membranes, and steroids.
How does Acid affect cell membranes?
And none of the cell’s activities would be possible without thin lipid membranes, or bilayers,that separate its parts and regulate their functions. … Changes in the packing of the tails into a hexagonal, rectangular-C, or rectangular-P lattice are observed at various pH levels.
What is an amphiphilic molecule attracted to?
Amphiphilic molecules is a general term that describes any compound that contains two distinct covalently bonded components with different affinity for the solvent in the same molecule, in which one part possesses a high affinity for polar solvents (such as water), and another part has a strong affinity for nonpolar …
What makes something amphoteric?
In chemistry, an amphoteric compound is a molecule or ion that can react both as an acid and as a base. … One type of amphoteric species are amphiprotic molecules, which can either donate or accept a proton (H+). This is what “amphoteric” means in Brønsted–Lowry acid–base theory.
Is amphoteric and Amphiprotic the same?
Amphoteric Versus Amphiprotic An amphoteric substance is one that can act as either an acid or a base. An amphiprotic substance can act as either a proton donor or a proton acceptor. … An example of an amphoteric compound that is not amphiprotic is ZnO, which can act as an acid even though it has no protons to donate.
What is an amphipathic protein and how does it help insert into a membrane?
Amphipathicity is the segregation of hydrophobic and hydrophilic amino acid residues between the two opposite faces of the protein α-helix, a distribution well suited for membrane binding (Drin and Antonny, 2010; Giménez-Andrés et al., 2018).
How is a lipid anchored protein attached to the membrane?
Lipid-anchored proteins (also known as lipid-linked proteins) are proteins located on the surface of the cell membrane that are covalently attached to lipids embedded within the cell membrane. These proteins insert and assume a place in the bilayer structure of the membrane alongside the similar fatty acid tails.
Why cholesterol is known as amphipathic molecules?
Cholesterol is referred to as an amphipathic molecule, that it contains its hydrophilic and hydrophobic parts. The hydroxyl group (-OH) in cholesterol is aligned with the phosphate head of the phospholipid on the cell membrane, which the rest of the cholesterol goes with the fatty acid of the membrane.
What is an amphipathic molecule quizlet?
amphipathic molecule. A molecule that has both a hydrophilic region and a hydrophobic region. aquaporin. A transport protein in the plasma membrane of a plant or animal cell that specifically facilitates the diffusion of water across the membrane (osmosis)
Why fatty acids are called amphipathic?
Fatty acids are thus amphipathic – the carboxylic acid is ionized at physiological pH (making it a negatively-charged carboxylate group) and interacts well with a polar solvent (the carboxylate group is hydrophilic, or water-loving), while the hydrocarbon chain is quite nonpolar, and contributes a hydrophobic effect.
Are lipids hydrophobic or hydrophilic?
Molecules such as proteins, nucleic acids, and carbohydrates have an affinity for water and are called hydrophilic (“water-loving”). Lipids, however, are hydrophobic (“water-fearing”).
Are lipids Amphipathic?
All of the lipid molecules in cell membranes are amphipathic (or amphiphilic)—that is, they have a hydrophilic (“water-loving”) or polar end and a hydrophobic (“water-fearing”) or nonpolar end. … These have a polar head group and two hydrophobic hydrocarbon tails.