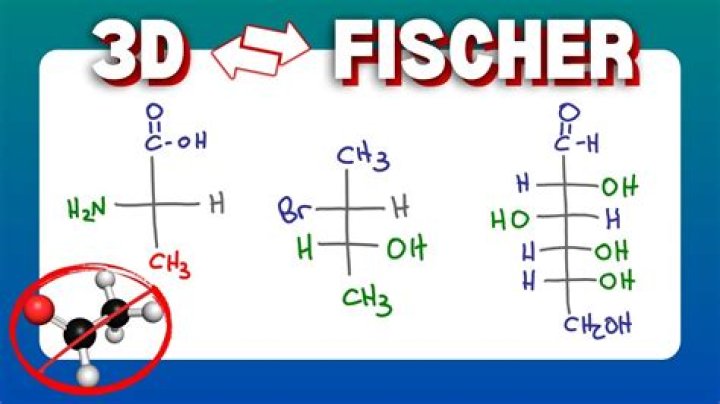
To make a Fischer projection, you view a chiral center so that two substituents are coming out of the plane at you, and two substituents are going back into the plane, as shown here. Then the chiral center becomes a cross on the Fischer projection. Every cross on a Fischer projection is a chiral center.
How do you know if something is chiral or not?
Test 1: Draw the mirror image of the molecule and see if the two molecules are the same or different. If they are different, then the molecule is chiral. If they are the same, then it is not chiral.
What are two ways of determining if a molecule object is chiral?
Look for carbons with four different groups attached to identify potential chiral centers. Draw your molecule with wedges and dashes and then draw a mirror image of the molecule. If the molecule in the mirror image is the same molecule, it is achiral. If they are different molecules, then it is chiral.
How do you know if Fischer projections are enantiomers?
To find the enantiomer of a molecule drawn as a Fischer projection, simply exchange the right and left horizontal bonds. To determine whether the molecule in Fischer projection is a meso compound, draw a horizontal line through the center of the molecule and determine whether the molecule is symmetric about that line.What is Fischer projection formula?
A Fischer projection or Fischer projection formula is a convention used to depict a stereoformula in two dimension without destroying the stereochemical information, i.e., absolute configuration, at chiral centers.
How can you tell if a Newman projection is meso?
Identification. If A is a meso compound, it should have two or more stereocenters, an internal plane, and the stereochemistry should be R and S. Look for an internal plane, or internal mirror, that lies in between the compound.
How do you look at a Newman projection?
Newman projections are drawn by looking directly along a particular bond in the system (here a C-C bond) and arranging the substituents so that they are equally spaced around the atoms at each end of that bond. The protocol requires that the atoms within the central bond are shown as a dot and circle as defined below.
How can you tell the difference between chiral and achiral?
A chiral object is not identical in all respects (i.e. superimposable) with its mirror image. An achiral object is identical with (superimposable on) its mirror image. Chiral objects have a “handedness”, for example, golf clubs, scissors, shoes and a corkscrew.How do you write a Newman projection?
In a Newman projection, we look lengthwise down a specific bond of interest – in this case, the carbon-carbon bond in ethane. We depict the ‘front’ atom as a dot, and the ‘back’ atom as a larger circle. The six carbon-hydrogen bonds are shown as solid lines protruding from the two carbons.
What makes a molecule chiral?Chiral molecules contain one or more chiral centers, which are almost always tetrahedral (sp3-hybridized) carbons with four different substituents. … A is not superimposable on its mirror image (B), thus by definition A is a chiral molecule.
Article first time published onHow do you know if a Fischer projection is D or L?
If the hydroxyl group (or amino group for amino acids) is pointing to the right in the Fischer Projection, the sugar (or amino acid) is designated as D. If the hydroxyl group (or amino group for amino acids) is pointing to the left in the Fischer projection, the sugar (or amino acid) is designated as L.
Are meso compounds chiral?
Meso compounds are achiral compounds that has multiple chiral centers. It is superimposed on its mirror image and is optically inactive despite its stereocenters.
What are chiral centers?
Definition of chiral center : an atom especially in an organic molecule that has four unique atoms or groups attached to it.
What is the difference between Fischer and Newman projections?
We’re starting with a Newman Projection in the staggered conformation. Which means the carbon in the front has an opposite orientation to the carbon behind it. On the Fischer Projection, the horizontal line represents the groups coming up and out of the page, which means they’re facing in the same direction.
How do you convert Fischer projection to zig zag?
about the conversion of a Fischer to zig-zag projection is that the placement of two adjacent groups (on the same side of the Fischer projection) to the oppo- site sides of the zig-zag back- bone can provide the correct zig-zag structure. This is true only if we are concerned with the relative stereochemistry.
How do you know if a molecule is D or L?
Step 1: Make sure that the molecule is drawn as the Fischer projection in which the carboxylic acid group is on top and the side chain on bottom. eg: Step 2: If the amine group is on the right of the carbon chain, assign the label D to the compound; if it is on the left of the carbon chain, assign the label L.
What are main features of Fischer projection?
The Fischer Projection consists of both horizontal and vertical lines, where the horizontal lines represent the atoms that are pointed toward the viewer while the vertical line represents atoms that are pointed away from the viewer.
What is the difference between a Fischer projection and a Haworth projection?
The key difference between Fischer projection and Haworth projection is that the Fischer projection shows the open chain structure of organic molecules, whereas the Haworth projection shows the closed-cyclic structure of organic molecules.
How do you know if a Newman projection is eclipsed or staggered?
When looking at a Newman projection, the dihedral angle is defined as the angle made between two designated atoms on the front and back carbons. Staggered and eclipsed refer the relative orientation of all the bonds on the front carbon versus all the bonds on the back carbon.
How do you tell if a Newman projection is staggered or eclipsed?
In an eclipsed conformation the carbons are aligned so that the hydrogens are lined up with each other. This creates steric hindrance between them. In a staggered conformation the atoms are all equally spaced from each other.
How do you know which Newman projection is least stable?
The CH₃-CH₃ eclipsed conformer is the least stable. That is the case as long as the CH₃ groups are within about 30 ° of each other. Then comes the CH₃-H eclipsed conformer (CH₃ groups at 120 °). It is still high-energy but more stable than the CH₃-CH eclipsed conformer.
What is the difference between achiral and meso?
The key difference between the terms achiral and meso is that achiral compounds have no chiral centers whereas meso compounds have multiple chiral centers. In summary, an achiral compound is the opposite of a chiral compound.
How do you determine Superimposability?
The most straightforward way to determine whether a given object is chiral is to draw or visualize the object’s mirror image and see if the two are identical (that is, superimposable). If the object contains an internal plane of symmetry then it must be achiral.
Which molecule is chiral?
A chiral molecule or ion exists in two stereoisomers that are mirror images of each other, called enantiomers; they are often distinguished as either “right-handed” or “left-handed” by their absolute configuration or some other criterion.
What is handedness or chirality?
Chirality is defined by the lack of certain features of symmetry, which lead to an object not being superimposable on its mirror image. Handedness is a different phenomenon relating to the ability to classify chiral objects into right-handed and left-handed objects.
What is difference between D and L?
Summary – L vs D Isomers The main difference between L and D isomers is in the position of –OH group in the penultimate carbon atom. In D isomer, the OH- group of the penultimate carbon is positioned on the right side whereas, in L isomer, the OH- group of the penultimate carbon is positioned on the left side.
What is L and D configuration?
A convention of nomenclature, devised in 1906, states that the form of glyceraldehyde whose asymmetrical carbon atom has a hydroxyl group projecting to the right is designated as of the d-configuration; that form, whose asymmetrical carbon atom has a hydroxyl group projecting to the left, is designated as l.